جامعة الكفيل
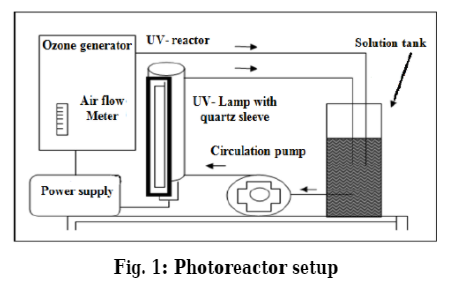
In this study, photo-ozonolysis has been used as advanced oxidation process for removal of color of dyes. The effect of concentration of dye, temperature, pH, flow rate of gas and light intensity has been studied. A complete removal has been achieved in a somewhat short period alternatively (60 min). The maximum removal of color adopted of high pH and temperature. The results have been shown that the rate of removal decreases with the increase of initial dye concentration, also it is found that the ratio of removal increases with increase of temperature. Moreover, one can notice from the results that with the increase of air flow rate and incident light intensity, the ratio of removal increases for all days used. The chemical oxygen demand (COD) was also studied in different conditions. The ratio of COD removal associates positively with the ratio of color removal for all dyes. The decolonization reaction was found to follow first order kinetics with respect to the dye concentration.

In this study, photo-ozonolysis has been used as advanced oxidation process for removal of color of dyes. The effect of concentration of dye, temperature, pH, flow rate of gas and light intensity has been studied. A complete removal has been achieved in a somewhat short period alternatively (60 min). The maximum removal of color adopted of high pH and temperature. The results have been shown that the rate of removal decreases with the increase of initial dye concentration, also it is found that the ratio of removal increases with increase of temperature. Moreover, one can notice from the results that with the increase of air flow rate and incident light intensity, the ratio of removal increases for all days used. The chemical oxygen demand (COD) was also studied in different conditions. The ratio of COD removal associates positively with the ratio of color removal for all dyes. The decolonization reaction was found to follow first order kinetics with respect to the dye concentration.